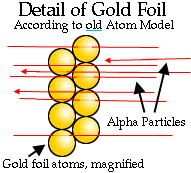
The point of this experiment was to justify the theory that atoms were permeable neutral balls. They expected that the high-energy charged alpha particles would have no problem zipping through a few atoms. The alpha particles should have just shot straight through the gold foil and left a small region at the back of the screen covered with dots.
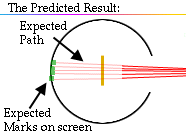
In fact, the screen showed dots mostly at the back of the screen; however, much to everyone's surprise, a few dots were scattered near the front of the screen.
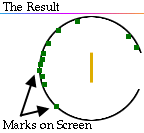
What could this mean?
The Standard Model Path includes an explanation of the atomic model in terms of subatomic particles.