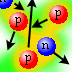
| Protons and neutrons migrate around inside a nucleus. |
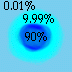
| So, you can consider the probability that a given proton or neutron is in a certain region. |
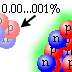
| There is a minuscule chance that a conglomeration of two protons and two neutrons (an alpha particle) may migrate outside the nucleus. There is a greater chance of this happening in a large nucleus than in a small one. |

| That alpha particle would be free of the residual strong force trapping it inside the nucleus, and like a suddenly released spring the charged alpha particle would "bounce" away from the nucleus. |
![]() This idea that "if it can happen, it will happen!" is fundamental to quantum
mechanics, the branch of physics that
explains particle behaviors in terms of
probabilities. For some atoms there is a certain
probability that it will undergo
radioactive decay due to the possibility
that the nucleus may --for the shortest
of instants-- exist in a state that allows it to blow apart.
This idea that "if it can happen, it will happen!" is fundamental to quantum
mechanics, the branch of physics that
explains particle behaviors in terms of
probabilities. For some atoms there is a certain
probability that it will undergo
radioactive decay due to the possibility
that the nucleus may --for the shortest
of instants-- exist in a state that allows it to blow apart.